The output density is given as kg/m, lb/ft, lb/gal (US liq) and sl/ft. Table E4: Specific Heat Capacity for Water C°(H 2O( l)) = 4. The calculator below can be used to calculate the ammonia gas density and specific weight at given temperatures and atmospheric pressure. While you can round the density to 1 gram per milliliter, there are more precise values for you to use. The usual value used in calculations is 1 gram per milliliter (1 g/ml) or 1 gram per cubic centimeter (1 g/cm 3 ). Table E3: Water K w and pK w at Different Temperatures (☌) Temperature The density of water is the weight of the water per its unit volume, which depends on the temperature of the water. Table E2: Water Vapor Pressure at Different Temperatures (☌) Temperature In this paragraph of heat of vaporization I got a bit confused by these numbers: 'Water’s heat of vaporization depends on the temperature: its around 540 cal/g at 100 ☌ (waters boiling point) and around 580 cal/g at 25 ☌ (room temperature). But you’re right that it does vary a little bit with temperature. Table E1: Water Density (g/mL) at Different Temperatures (☌) Temperature In general, we say that the density of water is 1000 kg/m3 (or 1 g/cm3). For example, the density of ultrapure water at 20.00 ☌ is known to be 0.998203 g/cm 3: any deviation from this value ± tolerances would imply that the water sample contains impurities. Round to three sig figs, the number of sig figs you have for the mass of the sample, the answer will be The measurement of density is crucial in different industries to ensure quality for both raw materials and finished goods. You can use the density of water as a conversion factor to determine what volume would #"9.98 g"# of water occupy at this temperature So, at water is said to have a density of #"0.998203 g mL"^(-1)#. The water density varies for different temperatures. I assume that the problem provided you with a table of water densities a various temperatures, but since you don't have it listed here, I'll use this resource to find it Density of Water at Various Temperature Scales. This means that you will get less than #"1.00 g"# per milliliter of water as you increase temperature. So, as temperature increases, the density of water decreases. Water has a maximum density of approximately #"1.00 g mL"^(-1)# at #-># you can read more about that in this Socratic answer.

The output pressure is given as kPa, bar, atm, psi (lb/in ) and psf (lb/ft.
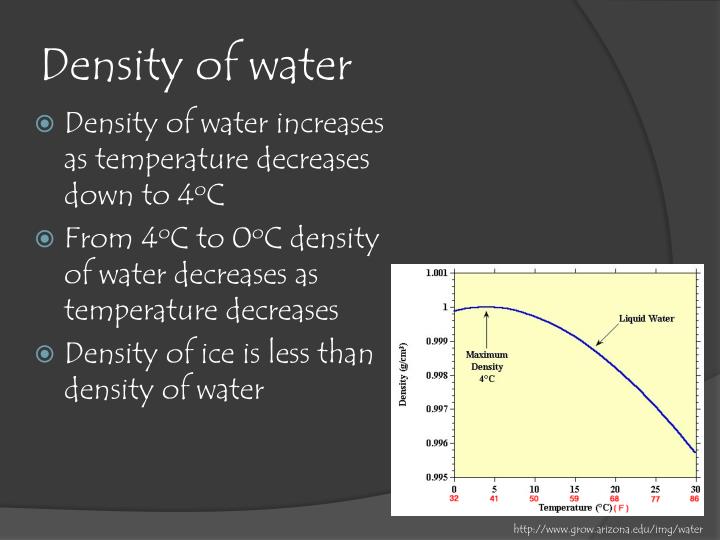
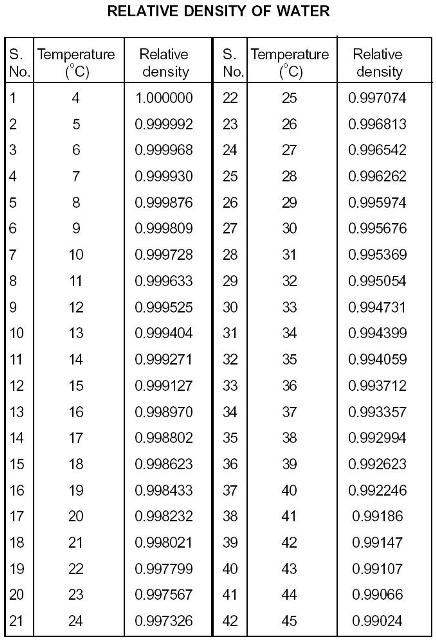
This is why the problem provides you with the temperature of the water. The calculator below can be used to calculate the water saturation pressure at given temperatures. The idea here is that water's density is temperature dependent, meaning that it varies depending on the temperature of the water sample.
